DriveAFM
Performance without compromise
The DriveAFM is the ultimate tool for nanoscale research. It allows you to explore a variety of phenomena in different domains. With the DriveAFM, you can visualize the DNA double helix, observe the complex patterns of 2D materials and their atomic structure, and measure the electronic properties of semiconductors with high precision.
The DriveAFM features CleanDrive, a photothermal cantilever excitation system that ensures optimal stability and performance. It also enables WaveMode, the fastest off-resonance mode on the market. The DriveAFM has a tip-scanning design that accommodates samples of various sizes and shapes without compromising its performance.
Speak with an Expert
Download the DriveAFM Brochure
The PDF brochure includes details on the DriveAFM's technology, application examples for different areas of interest and system specifications.

Performance Without Compromise
The DriveAFM, Nanosurf's revolutionary tip-scanning AFM system, empowers researchers with exceptional performance across a diverse range of applications, irrespective of the sample being analyzed. This unique, ultra-low noise architecture provides high-resolution data acquisition, unlocking unparalleled possibilities. It is compatible with high-fidelity small cantilevers that further boost performance.
Image: Atomic lattice of HOPG
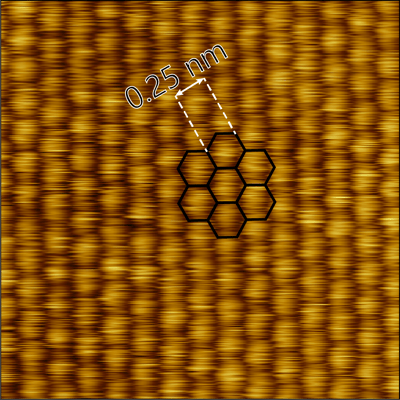
Versatility & Automation
No compromising on scan size or setup type. DriveAFM performs in a stand-alone setup as well as integrated with an inverted optical microscope. A full range of imaging modes, the possibility to use small cantilevers, and accessories and options covering virtually all imaginable research needs make DriveAFM future-proof and the best choice for demanding cutting-edge researchers.
The DriveAFM comes with full motorization that brings automation features that facilitate setting up and performing experiments.

Streamlined Operation with Intuitive Software
Nanosurf instruments have always been renowned for ease of use, and DriveAFM brings this to the next level with Nanosurf Studio: a control software designed from the ground up with the purpose of catering to the needs of demanding researchers, and making AFM more intuitively accessible to less experienced users.
DriveAFM for Life Science & Biotech
DriveAFM plays out all its advantages when it comes to dealing with measurements on biological samples. The full motorization allows you to control the system without interfering with a temperature-controlled environment, and seamless integration of the scan head with an inverted optical microscope allows transmitted light and fluorescence microscopy to be combined with AFM imaging and force spectroscopy. Benefit from correlative techniques and get results that drive forward your research.

High-Resolution Imaging
High-Resolution Imaging
DriveAFM is an optimal tool for high-resolution imaging of biological specimens, capable of capturing details as minute as the structure of single molecules. Its stable CleanDrive photothermal excitation feature ensures reliable performance, even in liquid conditions required to maintain biological sample integrity.
Furthermore, CleanDrive paves the path for WaveMode, the most rapid off-resonance imaging mode, enhancing the efficiency of imaging applications.
Image: Double-stranded DNA imaged in buffer solution revealing secondary structure of the DNA double helix. Image width: 110 nm
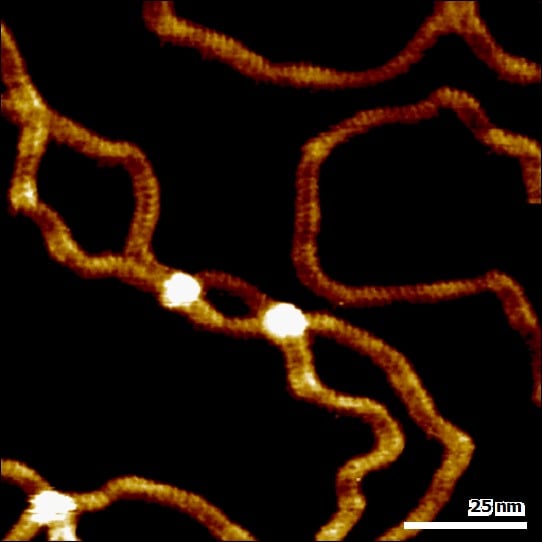
Most Gentle Imaging with WaveMode
Most Gentle Imaging with WaveMode
DriveAFM's WaveMode, powered by CleanDrive photothermal excitation, provides unprecedented speed in AFM off-resonance imaging without compromising "gentle touch" crucial for analyzing biological matter. This also enables researchers to investigate samples that before were hardly addressible with conventional AFM imaging modes, such as weakly attached samples or sensitive complexes. CleanDrive ensures the stable and reliable operation. Notably, the chosen 785 nm laser excitation and 840 nm deflection detection wavelengths are biocompatible and non-interfering with biological samples, even facilitating concurrent fluorescence imaging for comprehensive analysis.
Image: 3D topography of a single Herpes simplex virus type 1 (HSV-1) capsid revealing capsomere arrangement. Image width: 250 nm.
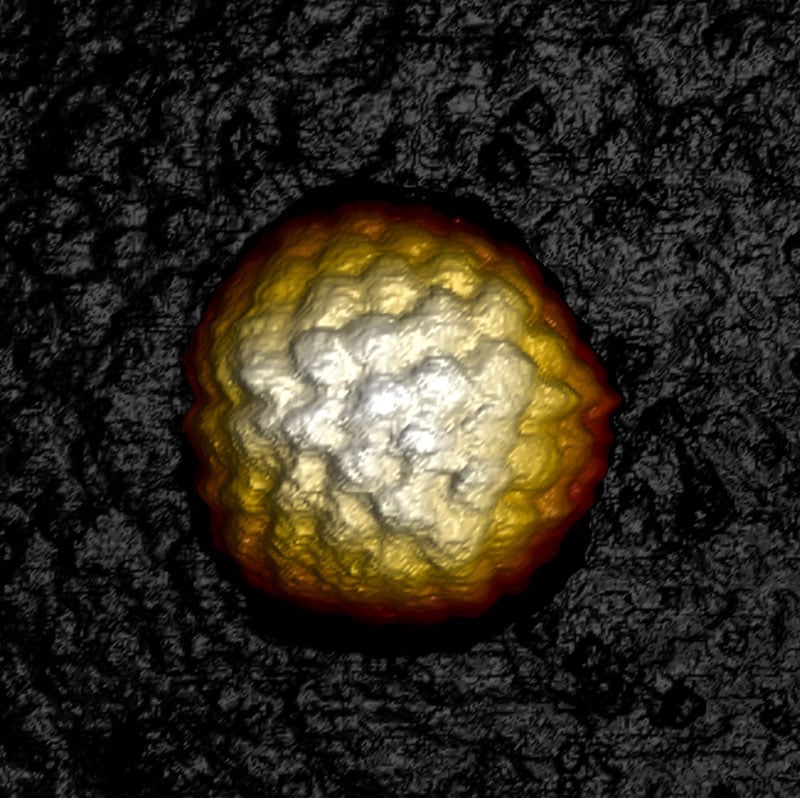
Nanomechanics at Your Fingertip
Nanomechanics at Your Fingertip
The Drive AFM provides a variety of methods to examine different aspecs of the nanomechanics of your sample, ranging from molecular adhesion or unfolding measurements to the mapping of nanomechanical properties and frequency-resolved dynamic mechanical analysis.
The Spectroscopy Plus feature empowers you to delve into force spectroscopy-based nanomechanical experiments. This versatile option opens up a range of possibilities, from basic force-distance curves to more intricate processes involving multiple indentation and relaxation cycles, revealing the viscous nature of your sample.
The newly introduced ViscoElastic Analysis option significantly broadens the frequency range for performing dynamic mechanical analysis. This facilitates the investigation of more localized dynamics, such as the relaxation modes of individual filaments within the cytoskeleton.
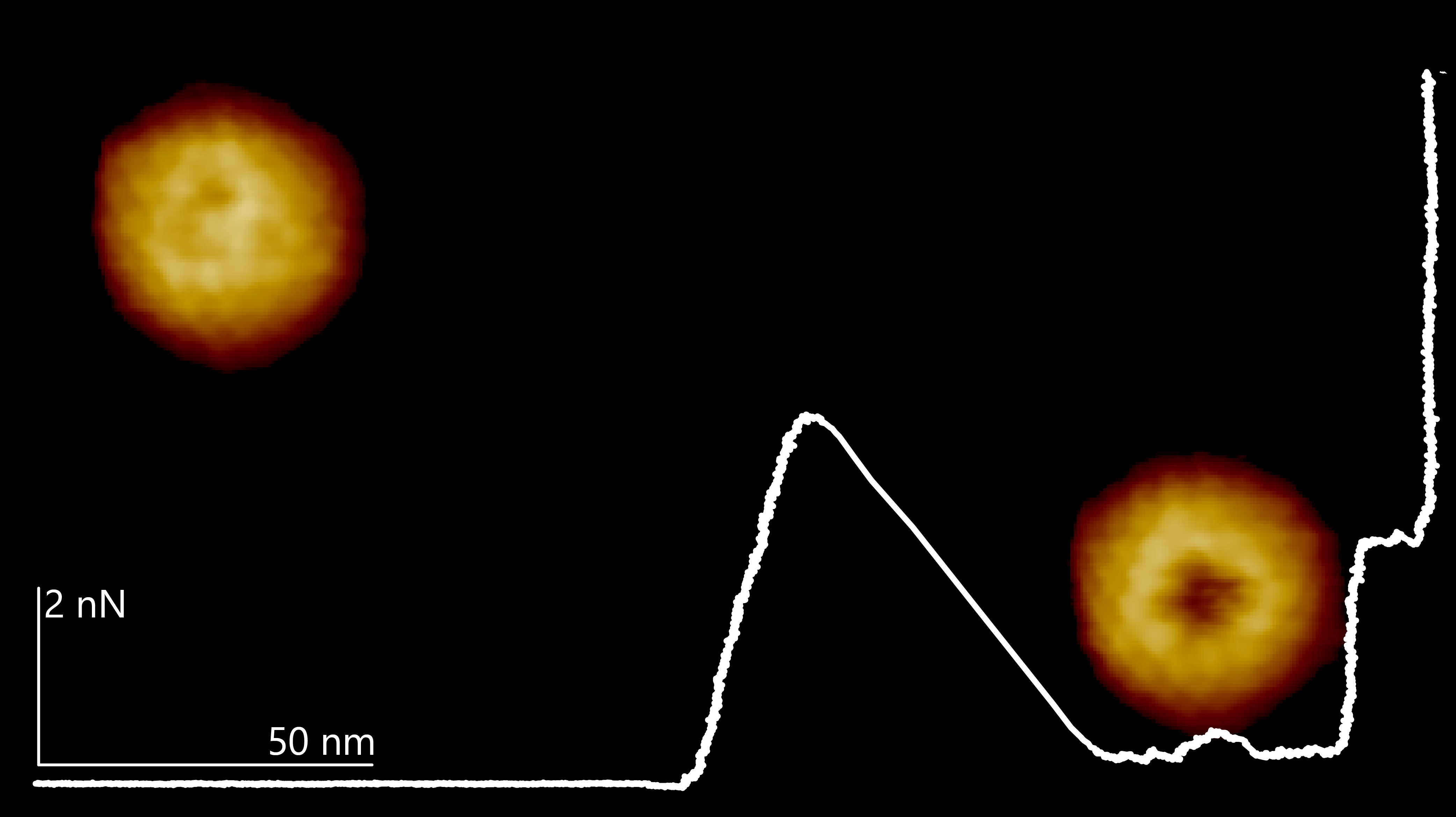
Image: Two HSV-1 capsids penetrated by an AFM cantilever tip. White line: Force distance curve showing indentation and penetration of the bottom right HSV-1 capsid from touching the upper capsid surface to full penetration.
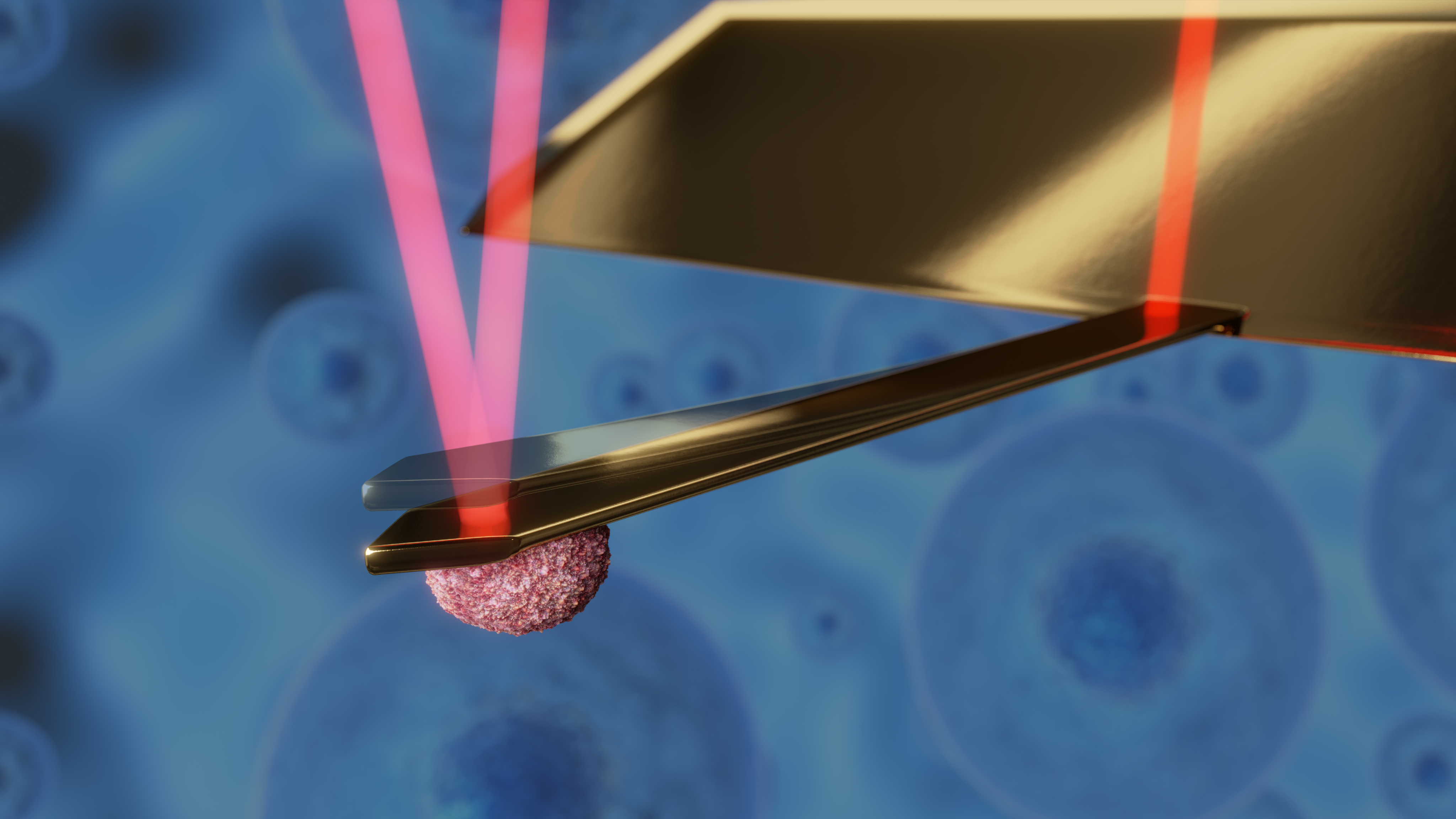
FluidFM
FluidFM
The DriveAFM is ready for research that goes beyond conventional AFM. FluidFM® combines microfluidics with the force sensitivity and positional accuracy of a Nanosurf AFM, unlocking exciting applications in single-cell biology and nanoscience. Conduct original research at the frontiers of science from bacterial adhesion to cell-cell interaction and from spotting to injection and extraction of cells. Benefit from optical, force, and fluidic control, with full software integration on DriveAFM. FluidFM can even be combined with PicoBalance to measure the mass of cells and microparticles picked up by the FluidFM probe.
Video: Injection of DAPI stain into the cytoplasm of a living cell.
PicoBalance
PicoBalance
PicoBalance enables users to non-invasively measure the mass of microscopic particles - e.g. cells – and track their mass over time. It is an integrated solution that combines mass monitoring with optical microscopy and long-term cell cultivation. PicoBalance allows novel research in the fields of cell mass or volume regulation, cell migration, cell nutrition, cell division and cell cycle progression, fat cell storage and metabolism, viral infection-related processes, ion channel properties, drug screening for drugs targeting pathways linked to cell growth, new therapies for cancer, aging, obesity, type 2 diabetes, neurodegeneration, and other diseases linked to a deregulation of growth control.
DriveAFM for Materials Research
The DriveAFM offers a combination of high performance and adaptability for a broad spectrum of applications in materials research. Its design ensures consistent and reliable operation across both air and liquid environments, and effectively handles large and heavy samples. The system is user-friendly, with automation features that streamline the process of conducting measurements on different parts of a sample.
Beyond basic imaging, the DriveAFM supports the investigation of a sample's electrical and mechanical properties at the nanoscale. It also accommodates a variety of accessories, enhancing its functionality to include temperature control, magnetic field application, electrical current detection, and the ability to observe dynamic changes in electrochemical processes.

WaveMode - Off-Resonance Imaging Redefined
WaveMode - Off-Resonance Imaging Redefined
WaveMode, a groundbreaking innovation by Nanosurf, revolutionizes off-resonance imaging by transforming the cantilever into a dual-function sensor and actuator. This unique design enables WaveMode to achieve unparalleled imaging speeds, making it the fastest off-resonance imaging mode available. With precise control over the tip-sample interaction, users can adjust the interaction force to meet the specific requirements of the sample, enhancing tip durability and allowing for comprehensive analysis under varying loads. The versatility of WaveMode makes it an essential tool for advanced surface analysis.
Video: Schematic illustration of WaveMode operation.
Glovebox-Ready
Glovebox-Ready
DriveAFM is perfectly suited for operation in a glovebox: full control of all mechanical adjustments via the software, a robust scan head with magnetic cantilever holders for easy operation even when wearing thick gloves. DriveAFM’s low-noise, high-fidelity nanoscale platform coupled with the most stringently controlled environment provided by the glovebox hands you everything you need for smooth glovebox operation. Your sensitive samples will be kept in optimal environmental conditions while you perform you AFM characterization.

Advanced Nanomechanical Characterization
Advanced Nanomechanical Characterization
The DriveAFM offers a comprehensive set of functionalities to explore the intricate nanomechanical properties of your materials. This includes quantifying adhesion forces, characterizing material response under deformation, and mapping spatial variations in mechanical behavior.
The Spectroscopy Plus module empowers in-depth nanomechanical investigations through force spectroscopy. It facilitates a wide range of experiments, from fundamental force-distance measurements to complex protocols involving multi-cycle indentation and relaxation. This enables the characterization of a material's viscoelastic behavior, providing insights into its energy dissipation and deformation mechanisms.
The newly introduced Viscoelastic Analysis module significantly expands the accessible frequency range for DMA measurements. This allows for the investigation of highly localized dynamic processes, such as the relaxation behavior of individual components within complex materials.
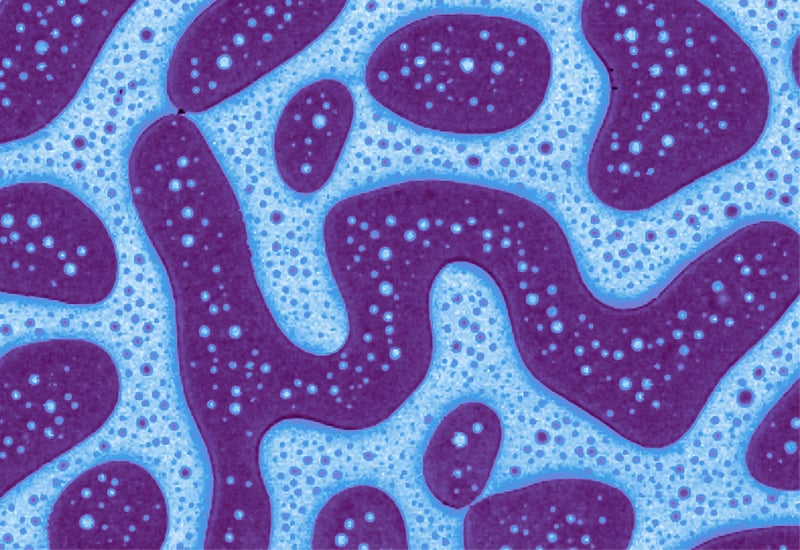
Image: Adhesion image of a SBS-PS thin film. Image width: 15.7 µm.
Nano-Electrical Characterization
Nano-Electrical Characterization
DriveAFM’s NEC features provide critical insights into the electrical behavior of materials at scales not visible to conventional microscopy techniques. From C-AFM to SMM, the full set of modes is available to analyze your samples. Quick and easy switching of setups for different types of measurements streamlines your measurement work and allows you to focus on what matters: achieving meaningful results.
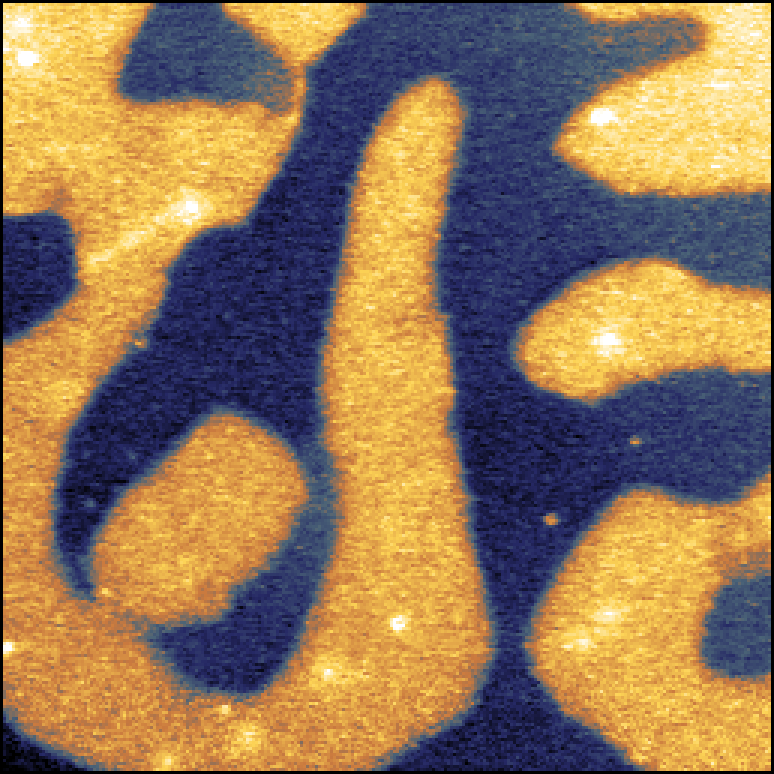
Images: Topography (left) and capacitance (right) images of a capacitance standard recodred using scanning microwave microscopy (SMM). Image widths: 52 µm.

DriveAFM for Electronics and Energy Materials
The DriveAFM stands out in the field of electronics and energy storage research, offering a glovebox-compatible solution that ensures precise AFM characterizations in controlled environments, crucial for sensitive samples. Its integrated SMM option and comprehensive NEC features provide a streamlined, user-friendly approach for in-depth nano-electrical characterization of materials, from dielectrics to metals. With minimal sample preparation, quick setup changes, and high-fidelity nanoscale measurements, the DriveAFM facilitates efficient and meaningful insights into the electrical properties of materials, enhancing research outcomes in these advanced sectors.

Glovebox ready
Glovebox ready
DriveAFM is perfectly suited for operation in a glovebox: full control of all mechanical adjustments via the software, a robust scan head with magnetic cantilever holders for easy operation even when wearing thick gloves. DriveAFM’s low-noise, high-fidelity nanoscale platform coupled with the most stringently controlled environment provided by the glovebox hands you everything you need for smooth glovebox operation. Your sensitive samples will be kept in optimal environmental conditions while you perform you AFM characterization.

Scanning Microwave Microscopy - Dissect the Dopant Profile
Scanning Microwave Microscopy - Dissect the Dopant Profile
Nanosurf's SMM Option is the fastest, most user-friendly solution for performing electrical characterization of dielectrics, semiconductors and metals with high sensitivity. It is set up in 3 clicks in the software, easy to operate and requires only minimal sample preparation. An added benefit is that both the AFM and SMM technology were developed at Nanosurf - resulting in better compatibility and much improved customer support over other solutions.
Images: Topography (left) and capacitance (right) images of a capacitance standard recodred using scanning microwave microscopy (SMM). Image widths: 52 µm.

Nano-Electrical Characterization
Nano-Electrical Characterization
DriveAFM’s NEC package provides critical insights into the electrical behavior of materials at scales not visible to conventional microscopy techniques. From C-AFM to SMM, the full set of modes is available to analyze your samples. Quick and easy switching of setups for different types of measurements streamlines your measurement work and allows you to focus on what matters: achieving meaningful results.
Image: Contact potential image of a BiSn alloy recorded using single-pass heterodyne KPFM. Image width: 5.5 µm.
DriveAFM Features
WaveMode
Photothermal off-resonance imaging offers the most gentle imaging available to date.
Small Cantilevers
Faster imaging compared to regular cantilevers.
PicoBalance & Viscoelastic Analysis
Gain more insights into your sample with mass measurements and frequency-resolved nanomechanics.
Motorization & Automation
Full motorization of the system allows automation of setup (e.g. cantilever alignment) and measurements
28-Bit CX Controller
High-resolution imaging across the full scan range.
Glovebox-Ready
Motorization and automation features facilitate experiments in highly controlled environments.
CleanDrive
Photothermal cantilever excitation - the most stable way to drive a cantilever.
Ultra-Low System Noise
The basis for investigating your sample in detail down to the nanometer.
FluidFM®
Cantilever-based microfluidics for spotting and deposition, single-cell manipulation and nanomechanics
DriveAFM Operating Modes
Standard imaging modes
- Static Force Mode
- Lateral Force Mode
- Dynamic Force Mode (Tapping Mode)
- Phase Imaging Mode
Advanced imaging modes
- CleanDrive photothermal excitation
- WaveMode
Electrical properties
- Conductive AFM (C-AFM)
- Scanning Spreading Resistance Microscopy (SSRM)
- Piezoelectric Force Microscopy (PFM)
- Electrostatic Force Microscopy (EFM)
- Kelvin Probe Force Microscopy (KPFM)
- Scanning Microwave Microscopy (SMM)
- Magnetic Force Microscopy (MFM)
Mechanical properties
- Force Spectroscopy (Unfolding and Stretching, Adhesion)
- Modular Spectroscopy (user-defined multi-segment force spectroscopy)
- Force Modulation
- Force Mapping (Stiffness and Modulus, Adhesion)
Lithography and Nanomanipulation
Electrochemical AFM (EC-AFM)
Scanning Thermal Microscopy (SThM)
Nanosurf Studio – Just Press Play
Our software engineers and application scientists took a good look at what performing atomic force microscopy measurements is actually like from a user perspective, and went back to the drawing board to create a user experience that allows you to focus on your measurement and your results. Look forward to a new release each quarter, with new features and improvements. Just press play.
- Set up your individual software GUI – for experts and novice users
- Automated AFM setup (laser alignments, spring constant and sensitivity calibration, approach, data acquisition)
- ViewPort optical sample navigation
- Quarterly releases with new features, improved workflows and bugfixes
- Easy to learn for novice users, all the flexibility for the expert user
- Instrument control is fully scriptable
Download the DriveAFM Brochure
The PDF brochure includes details on the DriveAFM's technology, application examples for different areas of interest and system specifications.

Accessories
#{ item.resourceType }
#{ item.date_text_field }
#{ item.name }
#{ truncateText(item.metadescription) }
#{ item.readmoretext }No posts available
Measurement Examples
#{ row.name }
There are no items to display.
Resources
#{ item.resourceType }
#{ item.date_text_field }
#{ item.name }
#{ truncateText(item.metadescription) }
#{ item.readmoretext }No resources available for these filters.
Interested in DriveAFM?
Get to know the DriveAFM! Reach out to us to discuss your application with one of our seasoned AFM experts, to get a budgetary quote or schedule a product demonstration or exploratory meeting.
